You probably know that generic drugs are just as effective as brand names. They have the same active ingredient, they're way cheaper, and they do the job. But there's something most people-and even some doctors-overlook: the stuff that isn't the medicine. Every pill contains excipients is inactive ingredients like fillers, binders, and preservatives that help a drug stay stable and easy to swallow. While one pill's fillers might be harmless, taking five or ten different generics can lead to a cumulative buildup of these substances, potentially triggering reactions your body can't handle.
Why "Inactive" Doesn't Mean "Inert"
It's a common misconception that inactive ingredients do nothing. In reality, they are functional components. For instance, lactose is a common sugar used as a filler in tablets. For most people, a tiny bit of lactose in a pill is no big deal. But if you're taking four different generic medications, each containing its own dose of lactose, you might suddenly find yourself dealing with severe gastrointestinal distress. You aren't reacting to the medicine; you're reacting to the filler.
The real problem is that the FDA is the federal agency responsible for protecting public health by ensuring the safety and efficacy of drugs only requires generics to be bioequivalent in terms of the inactive ingredient interactions and active drug performance. They don't require the inactive ingredients to match the brand name or other generics. This means every time you switch generic manufacturers, you're essentially swapping out the "recipe" of fillers in your medication.
The Danger of Cumulative Exposure
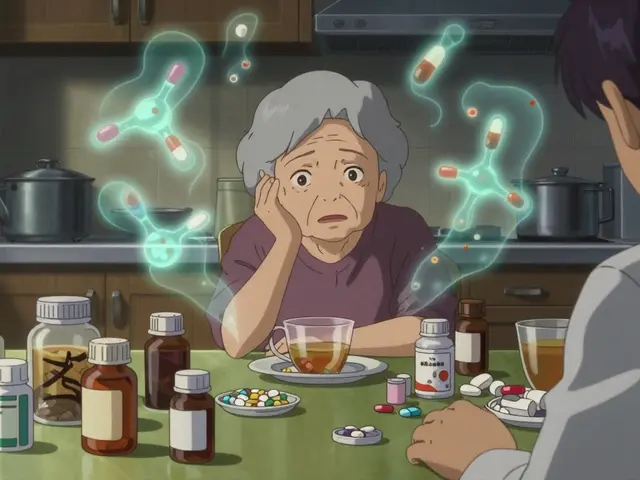
If you're managing a few health issues and taking several prescriptions-a situation known as polypharmacy-the math starts to add up. A study published in PubMed Central showed that a patient taking 10 medications could ingest nearly 3 grams of inactive ingredients every single day. When you're eating that much chemical filler, the risk of a "threshold event" increases.
Think of it like a bucket. Your body can handle a little bit of a specific dye or preservative. But as you add more medications, the bucket fills up. Eventually, one more pill pushes you over the edge, and you experience an allergic reaction or a stomach flare-up. This is why some patients report feeling fine on one generic, but suddenly develop hives or breathing issues after adding a third or fourth generic drug to their regimen.
| Inactive Ingredient | Typical Use | Potential Impact | Estimated Sensitivity |
|---|---|---|---|
| Lactose | Filler/Binder | Bloating, Diarrhea | ~65% of global population |
| Propylene Glycol | Solvent | Allergic reactions | Common in oral liquids |
| Tartrazine (Yellow 5) | Coloring agent | Skin rash, Asthma | ~4% of patients |
| Bisulfites | Preservative | Bronchospasm | 5-10% of asthmatics |

When Fillers Mess with Absorption
It's not just about allergies. In some cases, the choice of inactive ingredients can actually change how your body absorbs the active drug. This is a major concern for medications with a "narrow therapeutic index," where a tiny change in blood levels can mean the difference between a working dose and a toxic one. For example, the FDA has noted cases where patients switching between generic versions of digoxin experienced therapeutic failure because different excipients changed the drug's absorption rate.
Research has even shown a 15-20% difference in peak plasma concentrations for certain antiepileptic drugs when comparing brand-name versions to generics. If your medication is designed to prevent seizures, a 20% drop in effectiveness isn't just a nuance-it's a safety risk. This highlights why staying with the same generic manufacturer is often safer than switching for a slightly lower price.

How to Manage Your Generic Mix
Since the regulatory system doesn't automatically flag these interactions, the responsibility often falls on the patient and the pharmacist. If you have known sensitivities or are taking more than five medications, you can't just trust that "inactive" means "safe."
The best way to handle this is through a focused review of your medication list. You can use tools like DailyMed is a provider of official FDA-approved drug labels providing comprehensive information on ingredients to look up the full ingredient list for every pill you take. A pharmacist can help you calculate your daily intake of a specific filler, like lactose, to see if you're exceeding your tolerance threshold.
If you start feeling "off" after a pharmacy change or a new prescription, don't assume it's a side effect of the active drug. Ask your pharmacist: "Does this generic use the same fillers as my other medications?" Identifying one problematic dye or binder and switching to a different generic manufacturer can often resolve the issue entirely.
The Future of Ingredient Transparency
The industry is slowly waking up to this. The FDA's 2024 "Inactive Ingredient Transparency Initiative" is a step in the right direction, aiming to make all inactive ingredients available in digital labeling by the end of 2025. We're also seeing the rise of AI-powered tools like MedCheck AI, which can scan multiple prescriptions and flag potential excipient clashes with surprising accuracy.
Meanwhile, the European Medicines Agency (EMA) is already more strict, requiring manufacturers to justify the use of excipients that cause hypersensitivity in more than 0.1% of people. As our population ages and we take more pills to manage chronic conditions, this level of scrutiny will become a necessity rather than a luxury.
Are generic drugs less safe because of inactive ingredients?
No, they aren't generally "less safe," but they are different. Most people tolerate excipients perfectly. The risk arises primarily for people with specific allergies, severe intolerances (like lactose), or those taking a large number of different medications where the cumulative dose of a filler becomes a problem.
Why doesn't the FDA require generics to have the same inactive ingredients?
The FDA focuses on bioequivalence-meaning the active drug enters your bloodstream at the same rate and amount as the brand name. As long as the inactive ingredients are "acceptable" and don't interfere with the active drug's function, they are allowed to vary. This allows different companies to compete on price and manufacturing methods.
What should I do if I suspect a reaction to a filler?
First, keep a log of when the symptoms occur. Then, ask your pharmacist for the full ingredient list of the medication you recently started or switched. If you find a common allergen across multiple drugs, work with your healthcare provider to find alternative generic versions from different manufacturers that don't use that specific excipient.
Can inactive ingredients cause a drug to stop working?
In rare cases, yes. Some excipients can alter the absorption rate of the active ingredient. This is most dangerous in drugs with a narrow therapeutic index, such as digoxin or certain antiepileptics, where a small change in absorption can lead to therapeutic failure or toxicity.
How can I tell which generic manufacturer I am using?
Look at the prescription bottle or the packaging for the manufacturer's name. You can also check the NDC (National Drug Code) number on the bottle, which uniquely identifies the manufacturer and the specific formulation of the drug.